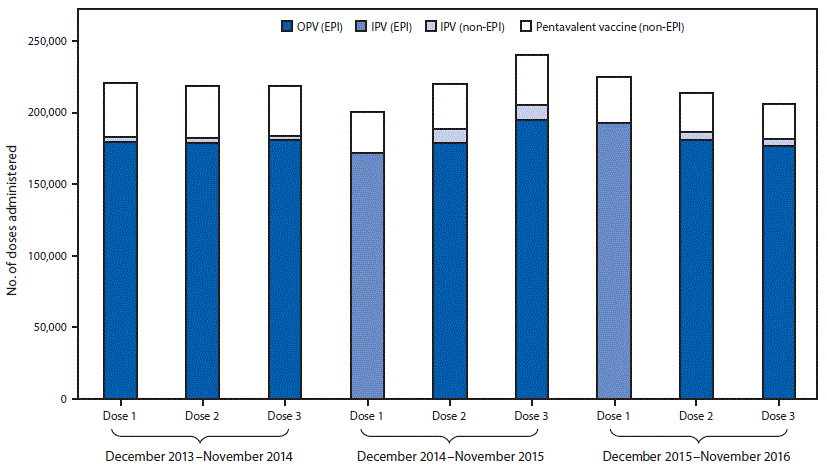
Introduction of Inactivated Poliovirus Vaccine and Impact on Vaccine-Associated Paralytic Poliomyelitis — Beijing, China, 2014–2016 | MMWR

Fractional dose compared with standard dose inactivated poliovirus vaccine in children: a systematic review and meta-analysis - The Lancet Infectious Diseases
Next Generation Inactivated Polio Vaccine Manufacturing to Support Post Polio-Eradication Biosafety Goals | PLOS ONE
Gavi, the Vaccine Alliance on Twitter: "Great news! Every country in the world – including all 73 Gavi-supported countries – have introduced inactivated polio vaccine (IPV). https://t.co/VaIFs5nV6L #EndPolio #IPV https://t.co/1JHXMucgcC" / Twitter

Preparation for global introduction of inactivated poliovirus vaccine: safety evidence from the US Vaccine Adverse Event Reporting System, 2000–12 - The Lancet Infectious Diseases

Inactivated Poliovirus Vaccine Made in Modular Facilities with Single-Use Technology - BioProcess InternationalBioProcess International

Recommendations to assure the quality, safety and efficacy of poliomyelitis vaccines (inactivated), Annex 3, TRS No 993